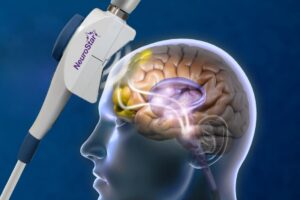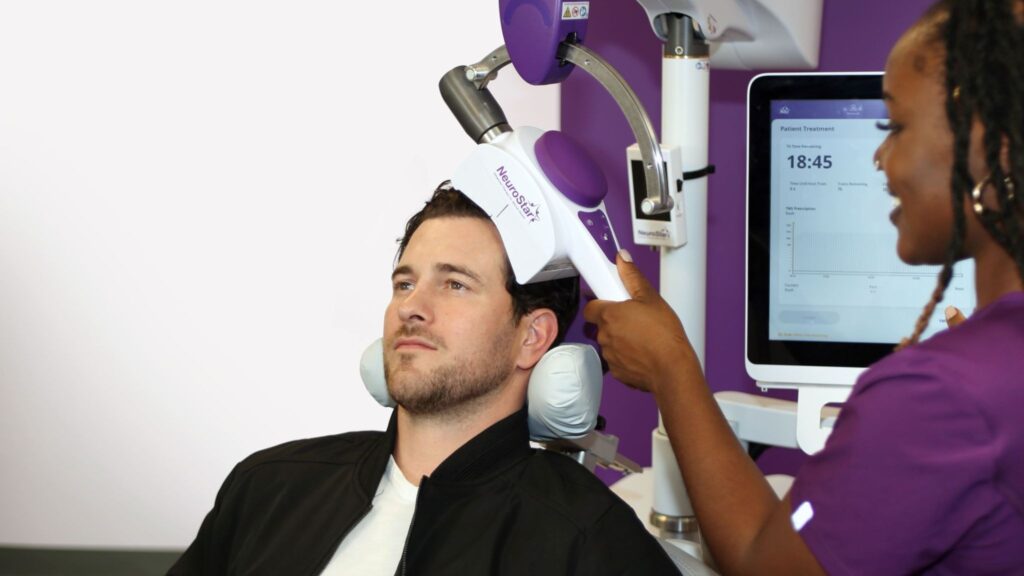
The FDA cleared Transcranial Magnetic Stimulation for treating major depression. This clearance is specifically for adults who didn’t get better after trying one antidepressant medication. It started in 2008 with the NeuroStar system, offering a different path when pills aren’t enough.
When antidepressants don’t relieve depression, transcranial magnetic stimulation (TMS) may be a next step, especially for those exploring structured options like TMS therapy for depression treatment. This FDA-cleared therapy uses magnetic pulses to stimulate mood-related brain areas.
We use NeuroStar at TMS of Tennessee. What does “FDA-cleared” really mean for you? What does it treat, and who is it for? Let’s get into the specifics.
TMS FDA Clearance: What You Need to Know
- FDA clearance for TMS and major depression began in 2008.
- Its primary use is for depression that hasn’t responded to medication.
- It’s a non-invasive procedure. Research indicates response rates can reach 83%.
What Is the Quick Answer About TMS FDA Clearance?
Yes, Transcranial Magnetic Stimulation is cleared by the FDA to treat major depressive disorder. This is specifically for patients who have not gotten better after trying antidepressant medications.
TMS is regulated as a medical device. That’s why the Food and Drug Administration uses the term FDA-cleared instead of FDA-approved. The clearance came through a standard review process that confirmed the treatment’s safety and that it works.
As noted by the Journal of Clinical Psychiatry
“Transcranial magnetic stimulation (TMS) is a US Food and Drug Administration-approved treatment for major depressive disorder (MDD) in patients who have not responded to 1 adequate antidepressant trial in the current episode.” – Psychiatrist.com
Here are the three main points:
- First clearance: 2008
- Main use: treatment-resistant depression
- A leading system: NeuroStar Advanced Therapy
The therapy uses a magnetic coil placed on the scalp. It sends magnetic pulses to specific areas of the brain that help regulate mood.
When Did the FDA Clear TMS for Depression?
The FDA first cleared TMS in 2008 for NeuroStar. Since then, the clearance has expanded to include more conditions and younger patients.
The path for Transcranial Magnetic Stimulation has been one of steady research. The first clearance was for adults with Major Depressive Disorder who hadn’t improved with at least one antidepressant. Later, more studies supported its use for other situations.
As highlighted by the American Psychological Association (APA)
“TMS is a noninvasive procedure that uses magnetic fields to stimulate nerves in the brain and was first approved by the FDA for the treatment of major depressive disorder (MDD) in 2008. Since then, technology has evolved… TMS has earned FDA approval for other conditions, including OCD, migraines, and smoking cessation.” – APA Services
Three major steps show this growth:
- 2008: Cleared for adult Major Depressive Disorder
- 2019: Cleared for anxious depression (depression with high anxiety)
- 2024–2025: Cleared for use in patients 15–21 years old as an add-on treatment
| Year | Milestone | Details |
| 2008 | First FDA Clearance | Approved for adults with Major Depressive Disorder (MDD) |
| 2019 | Expanded Indication | Cleared for anxious depression (MDD with anxiety symptoms) |
| 2024–2025 | Age Expansion | Cleared for patients aged 15–21 as adjunct treatment |
The studies that led to these decisions used common rating scales like the Hamilton Depression Rating Scale. Research in journals like Nature Mental Health shows these scales recorded real symptom improvement in patients getting repetitive transcranial magnetic stimulation (rTMS).
Other devices, like BrainsWay Deep TMS, have also been cleared. This shows that TMS devices are becoming a more established part of psychiatric care.
What Conditions Is TMS Officially Cleared to Treat?
TMS is officially cleared by the FDA for major depressive disorder in patients where antidepressants have failed. Using it for anything else is considered “off-label.”
The official uses for TMS therapy are limited to what the Food and Drug Administration has reviewed and confirmed. This is based on evidence from clinical trials.
The cleared uses right now are:
- Major Depressive Disorder (MDD)
- Treatment-resistant depression
- Obsessive-compulsive disorder (OCD)
- Anxious depression
However, doctors sometimes use TMS for other conditions. This is called “off-label” use. It’s not officially cleared by the FDA, but is legal when a doctor believes it may help. These include:
- Bipolar disorder
- Chronic pain
- Smoking cessation
It is important to know that for conditions like Parkinson’s Disease or Alzheimer’s Disease, TMS is not cleared and is still being researched.
According to the National Institute of Mental Health, off-label treatment requires a careful discussion between doctor and patient about the potential risks and benefits.
In our clinic, TMS of Tennessee, we follow FDA guidelines. We provide treatment only for its cleared uses, using the NeuroStar Advanced Therapy system.
How Does TMS Work for Depression?
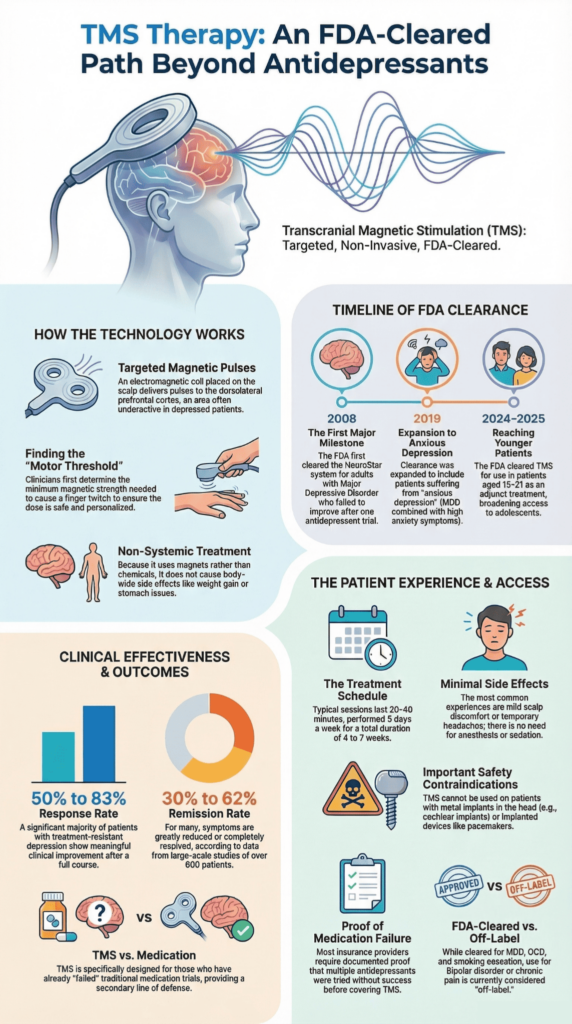
TMS uses magnetic pulses to stimulate a specific brain area, the dorsolateral prefrontal cortex. This region is often underactive in depression. A magnetic coil placed on the scalp generates a field that passes through the skull, activating nerve cells in that area.
Each session follows a set plan. We first find the right spot on your head and the right strength for the magnetic pulses.
A typical course of treatment looks like this:
- Session length: About 20 to 40 minutes
- Frequency: 5 days a week
- Total duration: 4 to 7 weeks
- An advanced option: Theta-burst stimulation (iTBS), which is a faster protocol often included in accelerated TMS treatment protocols
We start by finding your Motor Threshold. This is the minimum magnetic strength needed to cause a small finger twitch. It helps us set the correct dose for your brain safely.
According to Harvard Health Publishing, TMS helps improve how different parts of the brain communicate. It changes activity in the cerebral cortex to ease depression symptoms, all without putting medication into your whole body.
In our clinic, TMS of Tennessee, we use the NeuroStar Advanced Therapy system. It allows for precise targeting and gives real-time feedback during treatment.
How Effective Is TMS for Depression?

TMS is effective for many patients, with outcomes discussed in clinical detail in evidence on whether TMS works for depression, especially when viewed alongside broader comparisons like TMS Therapy vs Other Depression Treatments, which place its results in context with medication and other clinical options.
For those with treatment-resistant depression, studies show response rates between 50% and 83%. We track results using standard depression scales. Data from large studies, including those with the NeuroStar system, support this.
Key points about effectiveness:
- Designed for patients who haven’t improved with medication.
- Response (meaningful improvement) occurs in 50% to 83% of patients.
- Remission (symptoms go away) occurs in about 30% to 62%.
- A review of over 800 patients, including data from the VA Montana Health Care System, showed clear improvement after a full rTMS course.
- Combining TMS Therapy vs Other Depression Treatments can help more in complex cases.
| Outcome Type | Percentage Range | Meaning |
| Response Rate | 50% – 83% | Patients show significant improvement in depression symptoms |
| Remission Rate | 30% – 62% | Symptoms are greatly reduced or completely resolved |
| Study Size | 800+ patients | Based on large-scale clinical data including VA health systems |
At TMS of Tennessee, we use tools like the Hamilton Rating Scale of Depression to monitor progress.
Is TMS Safe and What Are the Side Effects?
TMS is generally safe. The most common side effect is mild scalp discomfort. Serious risks, like seizures, are very rare.
Safety was a priority for the FDA when clearing these devices. Years of clinical use confirm a strong safety profile compared to many medications.
Two things help explain the safety profile:
Most people feel some tapping or discomfort where the coil is placed. Any headache or discomfort usually goes away soon after the session ends.
No anesthesia is needed. This is different from treatments like Electroconvulsive Therapy (ECT), which has more procedural risks.
The side effects break down like this:
- Common: Scalp discomfort, headache
- Very Rare: Seizure risk (less than 1 in 1,000 patients)
- Not Present: There are no body-wide side effects like weight gain, sleepiness, or stomach issues.
There is one important safety rule. People with certain metal implants in the head (like cochlear implants) or implanted medical devices (like a pacemaker or defibrillator) cannot get TMS. The magnetic field could interfere with these devices.
At TMS of Tennessee, our safety check includes a full review of your medical history and any implants before we begin.
What Do Real Patients Say About TMS Therapy?

Patient stories vary. Many notice improvement after 10 to 36 sessions. Some see only partial help, and a few see little change.
Research studies give us the numbers, but listening to patients gives us the full picture. What people actually say highlights both the good and the not-so-good parts of treatment.
A couple of common experiences stand out.
Improvement is usually gradual. Most people don’t feel better after just one or two sessions. The change builds over several weeks.
The treatment itself can be uncomfortable. The magnetic coil makes a loud clicking sound and a tapping feeling on the scalp, which some find bothersome.
When patients talk about their experience, they often mention:
- Starting to feel better after 10 to 36 sessions.
- Feeling more interest in life and a lift in their overall mood.
- Dealing with some tiredness or increased worry in the first week or two.
It’s important to remember that results vary from person to person. This depends on how severe their depression was to start, if they attended all their sessions, and how their own brain responds. At TMS of Tennessee, we use regular check-ins and tools like the PHQ-9 questionnaire to track progress and make sure the treatment is on the right track.
What Are the Limitations and Insurance Challenges of TMS?

Getting TMS covered by insurance can be difficult. Companies often require proof that several antidepressant medications did not work.
Even if TMS is right for you, getting your insurance to pay for it is a separate challenge. Rules from Medicare and private insurers set strict conditions for coverage.
There are five main hurdles patients often face.
- Proof of Medication Failure: You typically need records showing you tried and did not improve on multiple antidepressants, often for an adequate amount of time.
- Limited Covered Diagnoses: Insurance usually only pays for FDA-cleared uses, like major depression. Using TMS for “off-label” conditions like generalized anxiety is rarely covered.
- Complex Paperwork: Getting approval requires specific diagnosis codes (ICD-10), detailed clinical notes, and correct billing forms.
- Regional Rules: Coverage can change depending on where you live. Medicare decisions are made by regional contractors, and their rules aren’t all the same.
- High Out-of-Pocket Cost: If insurance denies the claim, the full cost of treatment must be paid by the patient, which is a significant expense.
At TMS of Tennessee, we know this process is confusing. Our team helps by checking your insurance benefits upfront and handling the needed paperwork to support your case.
FAQ
Is Transcranial Magnetic Stimulation cleared by the Food and Drug Administration for depression?
Yes. Transcranial Magnetic Stimulation is cleared by the Food and Drug Administration for treating Major Depressive Disorder, particularly in individuals with treatment-resistant depression. TMS therapy uses magnetic pulses and controlled magnetic fields to influence brain activity in the dorsolateral prefrontal cortex.
Clearance requires premarket notification under 21 CFR 807.87(e), along with clinical testing data to confirm safety features and consistent treatment performance.
How does TMS therapy compare to Electroconvulsive Therapy for major depression treatment?
TMS therapy and Electroconvulsive Therapy both treat major depression, but they differ in method and side effect profile. TMS therapy uses a magnetic coil to deliver repetitive transcranial magnetic stimulation to the cerebral cortex, while Electroconvulsive Therapy uses controlled electrical currents under anesthesia.
TMS therapy does not require sedation and has a lower risk of memory-related side effects, making it suitable after medication resistance.
What conditions besides Major Depressive Disorder can TMS therapy help treat?
In addition to Major Depressive Disorder, TMS therapy is cleared for obsessive-compulsive disorder and is also used for smoking cessation. Ongoing research examines its role in other mental health conditions such as bipolar disorder, Parkinson’s Disease, and Alzheimer’s Disease.
Techniques like Deep TMS and theta-burst stimulation target the medial prefrontal cortex and anterior cingulate cortex to improve brain activity and support measurable clinical response.
Is TMS therapy safe for patients with implants or other medical devices?
TMS therapy is considered safe when proper screening is completed. Patients with metal implants, cochlear implants, or implantable cardioverter defibrillators require careful evaluation because magnetic fields may cause electromagnetic interference. Clinicians assess motor threshold, motor hotspot, and stimulation volume before treatment.
Modern TMS devices include built-in safety features, defined magnetic field characteristics, and seizure management protocols to ensure patient safety throughout treatment sessions.
What should patients expect during TMS treatment sessions for depression?
During TMS treatment sessions, a helmet-like coil is positioned on the scalp to deliver magnetic pulses to specific cortical targets. Each session typically lasts 20 to 40 minutes and is repeated over several weeks.
Providers may use repetitive TMS (rTMS) or deep transcranial magnetic stimulation depending on patient needs. Clinicians monitor progress using standardized tools such as the Hamilton Depression Rating Scale to evaluate clinical response and track relapse into depression.
Bottom Line on TMS FDA Clearance
You’ve tried to manage this, medication, rest, and pushing through hard days. It’s fair to ask if anything will truly help. TMS is FDA-cleared and has helped many people who did not improve with standard care. It is not a cure-all, but it offers a safe, focused option that targets mood-related brain activity.
Waiting often keeps people stuck longer than needed. You deserve steady progress, not just coping. If you’re ready, take a simple next step and TMS of Tennessee. A careful review can help you decide if this is the right path forward.
References
- https://www.psychiatrist.com/jcp/effectiveness-transcranial-magnetic-stimulation-clinical/
- https://www.apaservices.org/practice/business/technology/on-the-horizon/transcranial-magnetic-stimulation